UPDATED AUGUST 2017
This is a step forward – but just a small step.
Associate Health Minister Peter Dunne has announced Cannabidiol (CBD) will remain a controlled drug with restrictions expected to be lifted in September so that GPs and other doctors can prescribe CBD to patients without needing special approval.
SUMMARY:
- GPs and other doctors will be able to prescribe CBD products without needing special approval;
- CBD products must be GMP (pharma grade) but will be allowed trace contamination of up to 2% other cannabinoids, meaning botanically-derived CBD will be allowed;
- Challenges remain for filling any prescriptions for CBD: no products are currently approved in NZ, none can be made in NZ, and imported products cannot be stockpiled here;
- Patients with a prescription will not be allowed to order suitable CBD products from overseas – they must ask their prescriber or pharmacy to import it for them;
- Funding will remain an issue, with pharma-grade CBD products relatively expensive.
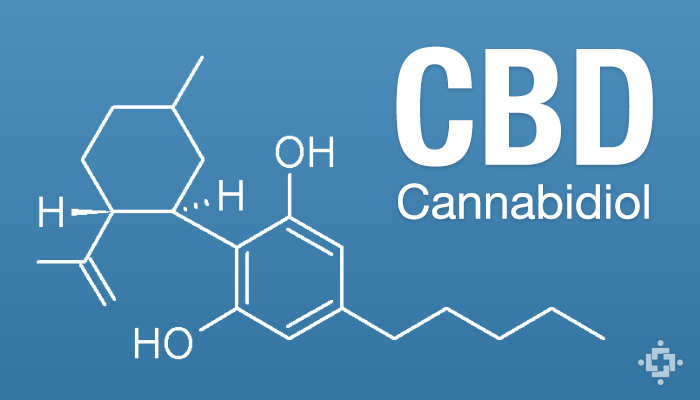
What is CBD and the current law?

CBD is a non-psychoactive cannabinoid found in cannabis plants that is indicated to help patients with a wide a variety of conditions including epilepsy, arthritis, cardiovascular disease, cancer, diabetes, gastrointestinal disorders, multiple sclerosis, PTSD, antibiotic-resistant infections, anxiety and other mental health disorders, and more. CBD can be made from legal hemp crops, and is available in many countries.
Despite not being listed in the Misuse of Drugs Act, the NZ Ministry of Health says CBD is a Class B1 controlled drug that has required special Ministerial approval under the 1977 Misuse of Drugs regulations.
Peter Dunne says the changes are “the culmination of a process which I began early this year after receiving independent advice from the Expert Advisory Committee on Drugs”. This was itself a result of Nelson lawyer Sue Grey’s threat of legal action against the Ministry of Health (supported by NORML), and the subsequent fast-tracking of a clinical trial that showed medical benefits from using CBD in children with epilepsy.
NORML believes these changes will help some patients – eventually – but more could be done. CBD will remain a controlled drug – and that means a lot. Although it will be easier to prescribe, there are no approved products. Manufacture in NZ will remain difficult if not impossible. Controlled drugs cannot be stockpiled from overseas either. Costs will remain an issue. But it is still a step forward, just a smaller step than we’d like.
MORE INFO:
- What is CBD? » https://www.projectcbd.org/about/what-cbd
- Comprehensive Guide To Cannabidiol » http://redeyesonline.net/cbd-cannabidiol/
READ PETER DUNNE’S ANNOUNCEMENT:
Peter Dunne 8 AUGUST, 2017
Changes to regulations will improve access to cannabidiol (CBD) products
Associate Health Minister Peter Dunne has today welcomed the removal of a range of controls on cannabidiol (CBD), calling the changes an important step in New Zealand’s continued implementation of a compassionate, innovative and proportionate drug policy.
The changes, to come into effect in early September will see CBD, a non-psychoactive component of cannabis, no longer subject to a range of conditions that other controlled drugs are required to adhere to.
“The Regulations’ appearance today at Executive Council, is the culmination of a process which I began early this year after receiving independent advice from the Expert Advisory Committee on Drugs”, says Mr Dunne.
“Following this advice, in May I took a paper to Cabinet arguing that the existing restrictions on CBD were no longer appropriate or justifiable.“
Today’s changes allow for a presence in CBD products of up to 2 percent of other cannabinoids usually found in cannabis, meaning trace amounts of THC, the psychoactive component of cannabis, will not be a barrier for people seeking to access CBD products.
Currently there is a limited range of CBD products made to a standard where prescribers can be sure the products contains what is claimed – and strict import and export restrictions on products sourced from other countries, which will continue to impact the supply of CBD products in New Zealand.
“In practical terms, the changes mean that as CBD products become available, doctors will be able to prescribe them to their patients in a similar way to any other prescription medicine.
“The changes reaffirm this Government’s commitment to an evidence based approach to cannabis-based products for medicinal purposes and future-proof access to such products.
“I am satisfied that CBD has potential therapeutic value and, as such, should be treated no differently to other medicines,” said Mr Dunne.
Further Information
Specifically, the Misuse of Drugs Amendment Regulations 2017 will:
- exempt CBD products with up to 2% of other cannabinoids usually found in cannabis from the requirement for Ministerial approval to prescribe;
- exempt pharmacies, medical practitioners, and wholesalers from the requirement to have an import licence for CBD products with up to 2% other cannabinoids;
- exempt CBD products with up to 2% other cannabinoids from the controlled drug records and stock keeping requirements;
- extend the allowable period of supply for CBD products with up to 2% other cannabinoids from one month to three;
- exempt CBD products with up to 2% other cannabinoids from the requirement to be prescribed on a triplicate form; and
- exempt specific controlled drugs that require refrigeration from the requirement to be stored in a safe in a pharmacy.
So what does this mean?
The Government’s intention is to pharmaceuticalise CBD, rather than let patients or caregivers grow their own CBD-rich herbal cannabis, or make it locally from hemp.
CBD will remain a controlled drug, but will be easier to prescribe.
Doctors will be able to prescribe approved products (when and if any are approved), or unapproved products manufactured overseas.
Patients with prescriptions will be able to buy their own from overseas and have up to three month’s supply sent to New Zealand, or they can have their prescription filled locally if their prescriber, pharmacy or a pharmaceutical company brings it in for them (controlled drugs are not allowed to be stockpiled).
NORML believes these changes will help some patients – eventually – but more could be done.
It’s good there will be a 2% tolerance for trace amounts of other cannabinoids, as this means that botanically-sourced (ie, natural) cannabis can be used rather than having to make CBD in a lab.
However it will remain very difficult to register products made from cannabis plants. Controlled drugs can only be manufactured here for research or study, not commercial purposes. Registering an approved product requires clinical trials, which could take years, and can only be conducted using GMP certified cannabis. Importing or distributing medical cannabis products requires permits which can only be obtained by research institutions or pharmaceutical companies.
Unapproved products will also need to be GMP-grade, or the prescribing doctor can expect close and ongoing scrutiny. For many doctors it will be too hard and too risky for them to prescribe unapproved products.
Funding will also remain an issue, with any pharma-CBD products expected to be expensive like Sativex. This would put it out of reach for many patients, especially compared to growing their own, or making it from hemp.
However if we took a herbal remedy or food-based approach, then local and affordable cannabis-based products could be available here within months.
- GIVE-A-LITTLE TO SUPPORT SUE GREY’S LEGAL CHALLENGE
- FEBRUARY 2017: Changes announced by Govt will make little difference, says NORML
- Specialists to prescribe medicinal cannabis products under Govt proposal

NEWS COVERAGE
- Peter Dunne press release, 8 August 2017: Changes to regulations will improve access to cannabidiol (CBD) products
- Peter Dunne press release, 2 June 2017: Government to ease restrictions on Cannabidiol
- Radio New Zealand, 2 June 2017: Doctors now able to prescribe cannabidiol
- Radio New Zealand, 2 June 2017: Govt loosens restrictions on medicinal cannabis
- TVNZ 1 News, 2 June 2017: Restrictions on medical cannabis product CBD to be removed
- Maori Television, 2 June 2017: Cannabis compound a prescription medicine in NZ
- Stuff.co.nz, 2 June 2017: Who will be able to get cannabidiol prescribed by a doctor – and when
- Newshub/Three, 2 June 2017: Medicinal cannabis restriction ease ‘not enough’
- Newstalk ZB, 2 June 2017: Cannabidiol won’t solve everyone’s problems, says doctor
- Newshub/Three, 3 June 2017: Cannabidiol great, but costs a lot – user
- Stuff.co.nz, 3 June 2017: Kiwis are literally dying to use medicinal cannabis. Is a change to the system bold enough?
APPLICATIONS SO FAR
Data supplied to Stuff by the Ministry of Health shows approvals of applications to use pharmaceutical-grade products – Sativex and Tilray – reaching a peak, at 59.
(SOURCE: Ministry of Health/Stuff.co.nz)
JANUARY 2017:
Nelson lawyer Sue Grey is pursuing legal action against the Ministry of Health over the legal status of cannabidiol (CBD).
- Papers obtained by NORML show the Ministry of Health is acting unlawfully and ignoring expert advice
- Dr Keith Bedford says CBD is not covered by the Misuse of Drugs Act, and does not require special approval
- NORML is supporting this action because sick people are being denied medicine and unnecessarily suffering
The current situation
Cannabidiol is a non-psychoactive cannabinoid widely used in Europe, Asia and the USA. There are high levels of CBD in hemp and some other cannabis strains. It does not get you high but it does have therapeutic benefits including reducing anxiety and depression, inflammation, and according to recent studies it even has promising anti-cancer properties.
One in twenty New Zealanders uses cannabis medicinally, and many of them are seeking the benefits obtained by CBD rather than THC.
LIKE THIS? DONATE ON GIVEALITTLE TO SUPPORT SUE GREY LAWYER
Unlike THC, Cannabidiol is not actually listed in the Misuse of Drugs Act. Yet the Ministry of Health claims it is a Class B1 controlled drug – the same as amphetamines, ecstasy and cocaine – because they say it is an isomer of THC and therefore captured by analogue provisions – and therefore the Misuse of Drugs Regulations 1977, section 22.
This means that even though CBD is listed as a prescribable medicine in the Medicines Act, doctors must jump through a lot of hoops to actually prescribe it, including asking Peter Dunne for special approval.
In the case of Alex Renton, that approval for Elixnol took months. An approval for a second product called Aseco also took months. No other approvals for CBD have been granted to date.
Customs, acting on orders from the Ministry of Health, are also seizing CBD at the border. But these heavy restrictions on the use and importation of CBD are in fact taking place outside the law.
The Ministry’s usual scientific advisor, Dr Keith Bedford of ESR, says that CBD is not an isomer, and that since it is not covered by the Misuse of Drugs Act any doctor can prescribe it without needing special approval.
Dr Bedford was for many years the Chief Analyst who was required by the Misuse of Drugs Act and the Medicines Act to identify and analyse drug samples, and is probably the most qualified person in the country on these matters. He has prepared an affidavit supporting the legal challenge.
Papers obtained by NORML show that rather than accept his advice, the Ministry has contorted itself trying to find something to support what they would like the law to say. Their policy is now based, it seems, on the advice of a secret scientist, whose name has been redacted.
This means patients are being denied access to CBD and unnecessarily suffering because the Ministry of Health is acting outside the law.
What we are seeking
Read the letter from Sue Grey to the Ministry of Health here.
The Ministry of Health policy could be amended overnight. They could accept that cannabidiol is not covered by the Misuse of Drugs Act, and tell Customs to stop seizing it. They have been arguing internally about this for at least three years. Cannabidiol (CBD) is not listed in the Misuse of Drugs Act, so does not require special approval. The Ministry of Health is acting unlawfully and ignoring expert advice.
What this would mean
CBD would be treated similar to codeine, which can be prescribed by doctors and also obtained over the counter, and also like poppy seeds which are widely used in foods.
For medicinal uses, any doctor could prescribe CBD to any patient, for any condition, without having to ask Peter Dunne for special approval. CBD medicines would need to be registered and approved, or a ‘manufacturing pharmacist’ could produce it locally.
For nutritional or wellness uses, CBD could be available in hemp foods and balms, without needing a visit to the doctor.
How you can help
Anyone who could potentially benefit from wider access to cannabidiol – patients, doctors, caregivers, friends and family, growers, processors and potential product makers – can support this:
- Donate to our legal costs: use NORML NZ Inc’s account (ASB) 12-3057-0594667-00
- Support media coverage with positive comments, letters to editors, coming forward with your own story, etc. Key messages to promote are:
- CBD is not psychoactive and could help a lot of people
- CBD is not actually listed in the Misuse of Drugs Act, so it does not require special approval
- The Ministry of Health is acting outside the law
- Sick people are suffering
- Get involved with the case by providing an Affidavit (which is like a witness statement – Sue Grey can help draft it)
Media coverage
Is the Ministry of Health acting outside the law on medical cannabis?
Hard News by Russell Brown – 11:40 Jan 16, 2017
The key barrier to the use of medical cannabis – or to even discovering what its uses might be – has long been marijuana’s illegality under the Misuse of Drugs Act. But what if it transpired that a key component of cannabis is not, and has never been, controlled by the act? And that heavy restrictions on its use and importation are in fact taking place outside the law?
This may sound to you like one of those conspiracy theories your embarrassing friend posts on Facebook, but it’s quite serious. And it’s the subject of an intended legal challenge notified by Nelson lawyer Sue Grey. You may recognise her name: she’s the lawyer who won Rebecca Rieder the right to walk through New Zealand Customs with a jar of raw cannabis prescribed to her in Hawaii.
Grey’s intention, and its legal and scientific basis, is summarised in a letter sent to the Ministers of Health and Customs at the beginning of this month. I’ve uploaded the letter here – it’s admirably clear in its language and worth reading.
…
Although Sue Grey is acting on behalf of a client, her action is being supported by a wider group of advocates, caregivers, processors and growers, along with The Hemp Store and Norml NZ. Norml president Chris Fowlie, who told me:
“CBD could improve the lives of many people. It is not psychoactive, has no known side effects, and is widely used in the USA and Europe. They say we must follow the law. We just want them to follow the law too.
“CBD is not listed in the Misuse of Drugs Act, and we believe the Ministry of Health and Customs are acting unlawfully when they seize it, and when they tell doctors they need special Ministerial approval to prescribe it. In fact, any doctor can prescribe it to any patient, for any condition.
“It seems extraordinary that officials have disregarded advice of their own expert, Dr Keith Bedford, and have instead based policy on the opinion of a secret scientist, whose identity and qualifications they have redacted. This is unprecedented in public policy, and we believe it will not stand up in court.”
Nelson Mail: Nelson lawyer Sue Grey takes government to High Court over cannabidiol
NINA HINDMARSH
Last updated 07:49, January 21 2017
MARION VAN DIJK –Nelson public rights lawyer Sue Grey.Sick New Zealanders might not need to jump through so many hoops to obtain some cannabis-based medicines if a Nelson lawyer wins her battle against the Government.
Sue Grey says she has the backing of a leading government scientist who says CBD based medicines should not be restricted because CBD isn’t covered under the Misuse of Drugs Act
Grey intends to challenge the Government in the High Court if it doesn’t change its legal classification of cannabidiol, or CBD.
Sunday Star-Times: Women of Influence
Great-grandmother battles Ministry of Health over medicinal cannabis productsCRAIG HOYLE
Last updated 05:00, January 22 2017
DAVID WHITE/FAIRFAX NZ – Schomburg suffers from chronic pain brought on by RSI and rheumatoid arthritisAn Auckland great-grandmother hopes an upcoming legal challenge will clear her and other medicinal cannabis users from being described as criminals.
Pearl Schomburg is one of many Kiwis who have found themselves on the wrong side of the law as the Ministry of Health argues with its own experts over how to classify cannabis extracts.
TVNZ: Stoush brewing between patients, Health Ministry over difficulty in accessing medical marijuana
Mon 23 January 2017
The Ministry of Health is being accused of acting outside the law by restricting some drugs from coming into New Zealand, when government scientists say they should be available through a doctor.
The Ministry has restricted some drugs from coming into NZ, but government scientists say they should be available through a doctor.
Radio New Zealand: Govt stance on medical cannabis ‘unlawful’
Nine to Noon, 11:36 am on 25 January 2017
People with chronic pain are being denied relief by an incorrect Health Ministry claim that a cannabis compound is covered by the Misuse of Drugs Act, a Nelson lawyer says.
CBD can help with nausea, sleeplessness and chronic pain, advocates say. Photo: 123rf
The Ministry of Health has said that cannabidiol (CBD), which is found in cannabis and hemp, is covered by the Act and has instructed Customs to seize any products containing it at the border.
Little Venus was suffering constant seizures until her mother ordered the banned substance online out of desperation.
By the time Ngaio gave her 18-month-old baby her first hit of cannabis oil, she felt she had nothing to lose. Seizures were gripping baby Venus every other day, becoming more aggressive each time. “You just feel so powerless” Ngaio tells VICE. “Nessie would double over like she’d been punched in the stomach. She was terrified. You never want to see your child look at you like that”.
Vice.com: WEED
Is Cannabis Oil CBD Actually Legal in New Zealand?Jan 25 2017, 3:26pm
Nelson lawyer Sue Grey says yes, and she is willing to take the Government to court to prove it.
(Top image via Flickr)
For people who are grappling with any variety of illness, cannabis-derived oil cannabidiol (CBD) has been hailed around the world as a miracle pain-relief alternative, due to its lack of side effects. It is legal in 40 countries and not listed in the United Nations treaty convention on Psychotropic Substances.
But access to CBD here is highly restricted. Sick New Zealanders, like toddler Venus, are forced to apply to the Ministry of Health, which can be costly, long, and often impossible. According to Nelson lawyer Sue Grey, for every 100 applications to the Minister, only 10 get approval. Sue says that those who’ve managed to avoid the system and get their hands on the drugs might be doing so illegally in the eyes of the Government—but she argues they’re acting legally under the rule of law.